Telehealth Abortion Faces A Federal Crackdown
Telehealth Abortion Faces A Federal Crackdown
Telehealth abortion access is suddenly on a knife-edge after a Louisiana federal court thrust mifepristone and cross-state prescribing into legal limbo, exposing how fragile digital care becomes when policy whiplash collides with patient needs. For patients and providers who bet on remote care to sidestep hostile local laws, the ruling reads like a warning shot: the convenience of doorstep delivery meets the brute force of courtroom politics. Investors eyeing digital health now have to reconcile growth projections with a patchwork of bans, while clinicians must navigate compliance risk that could chill innovation. The mainKeyword here is telehealth abortion access, and the tension between medical standards and judicial activism is shaping the next phase of reproductive care. The stakes are immediate: timelines for medication, safety protocols, and trust in virtual medicine all hinge on how this case evolves.
- Louisiana court scrutiny of
mifepristoneexposes telehealth abortion access to new legal friction. - Cross-state prescribing faces compliance risk that could slow digital health expansion.
- Patients may confront delivery delays, privacy concerns, and higher costs if restrictions tighten.
- Health startups must harden logistics, legal playbooks, and patient support to stay viable.
Why This Louisiana Case Matters For Telehealth Abortion Access
The ruling is more than a local skirmish. It tests whether federal approvals of mifepristone and misoprostol can be second-guessed by district courts, and whether telehealth prescribing that crosses state lines can survive a surge of injunctions. The mainKeyword is embedded in the conflict: telehealth abortion access depends on consistent federal backing and reliable shipping. When a single jurisdiction injects uncertainty, pharmacy partners hesitate, insurers recalculate, and patients shoulder the fallout. That ripple effect extends to broader digital health, where remote prescribing of controlled substances or sensitive medications now looks less predictable.
Key insight: A patchwork of state restrictions can freeze innovation faster than any technical bottleneck, because compliance risk dictates which features launch and which markets open.
The court challenge arrives as medical groups reiterate that mifepristone has decades of safety data. Yet legal claims argue process flaws and seek tighter controls. The result is a clash between evidence-based medicine and procedural litigation – a dynamic that often leaves patients stranded in the middle.
Interstate Friction And Platform Risk
Telehealth platforms built for scale now face the friction of fragmented compliance. If a ruling chills prescriptions into or out of Louisiana, neighboring states with similar laws might follow suit. That could force platforms to geofence services, throttle shipping, or require in-state clinicians, reducing the elasticity that made remote care attractive.
Providers are already modeling contingencies: rerouting fulfillment centers, maintaining parallel protocols for restrictive states, and investing in legal defense. Each layer adds cost and latency, eroding the core promise of rapid, discreet access.
How Providers Are Responding To The Crackdown
Clinicians and digital health operators are not standing still. Many are tightening documentation, strengthening patient identity verification, and refining decision trees to ensure prescriptions align with evolving rules. Some are exploring hybrid models that blend telehealth consults with in-person pickup to skirt shipping bottlenecks. Others are partnering with out-of-state pharmacies that can assume part of the regulatory risk.
Operational Playbooks Under Pressure
Expect to see updated clinical pathways that capture eligibility, consent, and follow-up with more rigor. Platforms may introduce hold periods to validate addresses and jurisdiction, even at the expense of speed. While that slows care, it reduces exposure to claims of improper dispensing.
Insurance dynamics will change too. Payers may balk at reimbursing services tied to contested medications, pushing more patients into self-pay. That cost shift could depress demand and widen disparities, especially for low-income patients who turned to telehealth precisely because it reduced travel and time off work.
Pro Tips For Patients Navigating Telehealth Abortion Access
- Confirm your state eligibility before booking: check if telehealth prescriptions for
mifepristoneormisoprostolare currently allowed. - Ask providers about shipping timelines and backup options like local pickup in case deliveries stall.
- Request clear aftercare instructions and emergency contacts; save them offline for quick reference.
- Use secure channels for communication; avoid sharing medical details over unsecured email.
Patients should also ask whether platforms maintain alternative fulfillment partners to reroute deliveries if a court order takes effect. Contingency planning is no longer optional – it is part of responsible care.
Why Investors And Founders Should Care
Digital health valuations have leaned on the assumption that regulatory risk is knowable. This case disrupts that math. When a district court can disrupt a medication with longstanding approval, the perceived stability of FDA-backed products erodes. Startups must budget for legal monitoring, rapid policy updates, and PR risk if service availability changes overnight.
Founders should treat compliance like core infrastructure. Building modular policy engines that toggle state-specific rules can prevent engineering fire drills. Investing in pharmacist partnerships and diversified shipping nodes reduces single-point failures. It is a reminder that healthcare innovation lives at the intersection of code, logistics, and law.
Investor takeaway: Growth stories that ignore regulatory volatility are incomplete; resilience against abrupt policy shifts now separates durable companies from speculative plays.
Main Scenarios To Watch Next
Scenario 1: Injunction Tightens Access
If the court issues a broader injunction on mifepristone, telehealth providers may pause services in affected states. Patients would face longer waits, and some might pivot to less regulated channels, raising safety risks. Pharmacies could limit stock to avoid entanglement, slowing the entire supply chain.
Scenario 2: Higher Courts Step In
An appellate stay could temporarily restore stability. Yet even a brief disruption would leave scars: insurers might renegotiate coverage, and platforms would keep stricter compliance mechanisms in place. Trust is easier to break than to rebuild.
Scenario 3: Legislative Counters
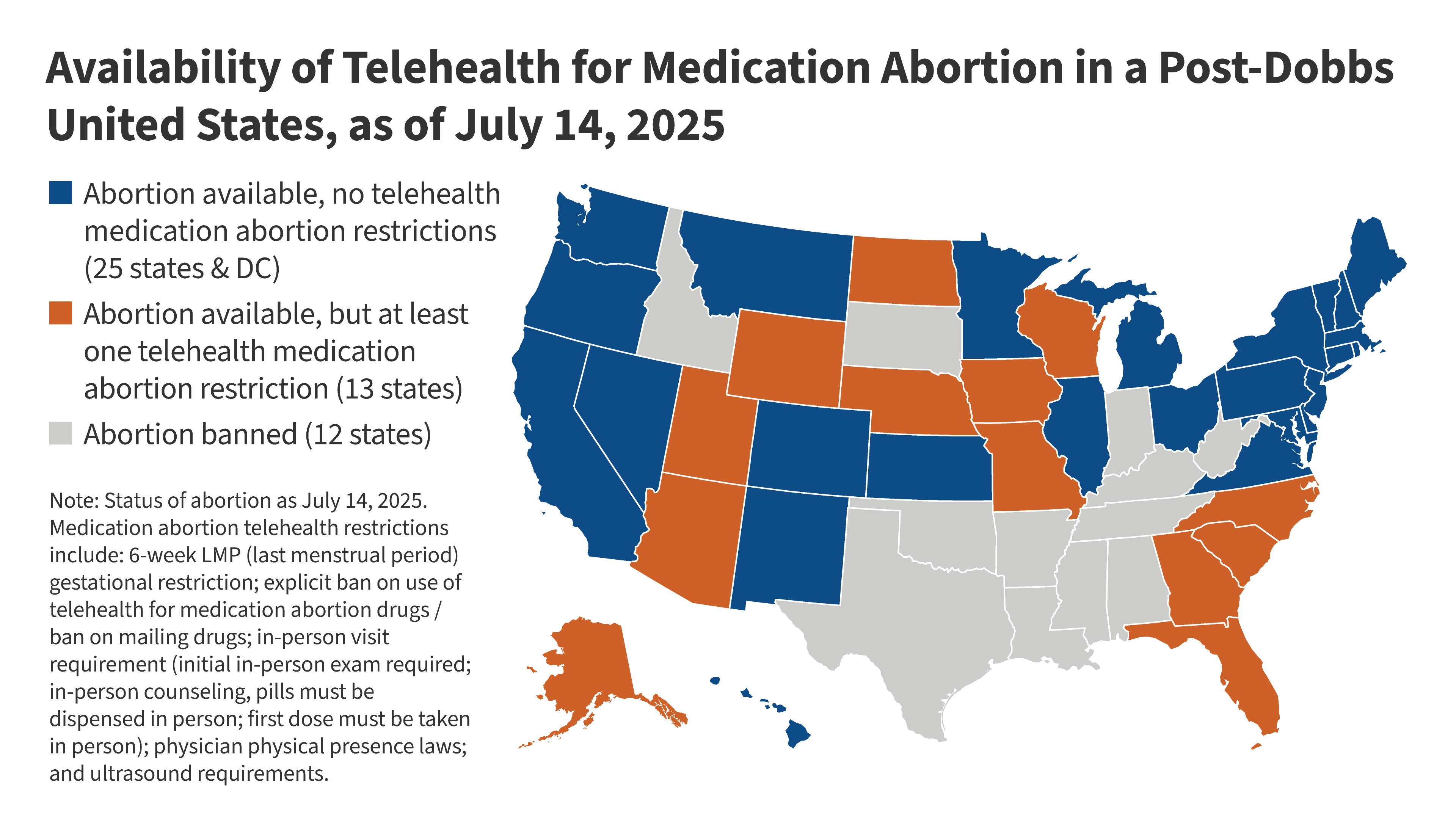
States supportive of reproductive care may pass shield laws that protect clinicians who prescribe across borders. That could create dueling legal frameworks and spark venue fights about where a case should be heard. For telehealth abortion access, that means a map of green and red zones rather than uniform availability.
Practical Steps For Telehealth Teams
- Audit workflows: Map every step from consult to delivery to identify where state law intersects with platform logic.
- Segment services: Use feature flags to disable or modify prescribing in response to rapid legal changes.
- Harden support: Train patient support teams to explain delays, refunds, or alternative care routes with clarity.
- Document evidence: Maintain clinical justifications and adherence records to counter claims of improper prescribing.
- Prepare comms: Draft templated updates for patients, partners, and media to deploy if access shifts.
These steps are not nice-to-have. They are survival tactics as regulatory headwinds intensify.
The Broader Implications For Digital Health
Telehealth flourished by stripping away geography. The Louisiana case reimposes borders digitally, reminding the sector that healthcare is never fully virtual. Expect renewed debates over the scope of federal preemption, the role of the FDA in defending its approvals, and whether courts should intervene in medication access absent safety concerns.
The ruling also tests public trust. Patients embraced remote care during the pandemic; they now need reassurance that legal turmoil will not interrupt critical services. How platforms communicate, comply, and adapt will set precedents for other sensitive therapies delivered through telehealth, from HIV PrEP to ADHD medications.
Future Outlook: Resilience Will Define Leaders
Assume volatility is the norm. The companies that win will be those that invest in compliance automation, diversify pharmacy relationships, and advocate publicly for stable regulatory frameworks. They will treat legal risk as a design constraint, not an afterthought. Patients will gravitate to providers that demonstrate transparency, reliability, and robust aftercare.
Telehealth abortion access stands at the crossroads of technology, healthcare, and policy. The Louisiana court may have fired the latest shot, but the battle will play out across appellate courts, legislatures, and the market itself. For now, the message is clear: speed and convenience must be matched by resilience and trust if digital reproductive care is to remain viable.
The information provided in this article is for general informational purposes only. While we strive for accuracy, we make no guarantees about the completeness or reliability of the content. Always verify important information through official or multiple sources before making decisions.